1. photograph clicker unit.
2. publish schematic.
3. enjoy the fun of graphs!
http://www.lib.uchicago.edu/~dean/MDWtoLAX-radiation.gif
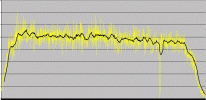
Southwest Airlines flight to LAX. I turned on the detector at roughly 12,000ft, and turned it off at the official 10,000ft announcement. According to the pilot cruising altitude was at 39,000ft. The big drop at 3/4 of the way across the graph was a temporary disconnection.
http://www.lib.uchicago.edu/~dean/radon15min.gif
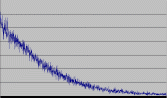
This is the decay of radioactive daughter products of radon-222 being captured on a coffee filter that filtered 15 minutes of air through a vacuum cleaner in someone's basement in Ohio.
If you graph this curve on a logarithmic scale, you get this:
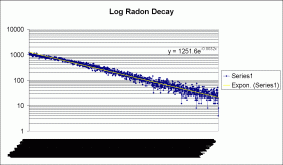
It's not quite straight, but the slope of the line gives you the exponent of the equation far below. The unreadable timescale is the same as the graph above it.
Radon-222-> Po-218 + alpha
Polonium-218-> Pb-214 + alpha
Lead-214-> Bi-214 + e-
Bismuth-214-> Po-214 + e-
Polonium-214-> Pb-210 + alpha
Questions I have that I haven't answered: What exactly is the software recording? What are those numbers? If I listen to the pulses, it seems the software multiplies the number of pulses by 4 to get the observed numbers, which it claims are microRads/hr. If this were a singular nuclear decay, I could deal with it, but it's 5 different decays. So how do I convert into pCi/L of radon? Why does it appear the half-life of the graph is nearly 50 minutes, which just happens to be the half-lifes of the first five decay products added together? Am I recording both the alpha particles and electrons/positrons, AND the gamma rays?
http://hps.org/publicinformation/ate/cat1.html
Simple EPA primer on radon: http://www.epa.gov/radiation/radionuclides/radon.htm
Uranium-238 decay chain: http://www.atral.com/U2381.html
Radioactive decay follows A=A(o)e^-kt, where A(0) is the inital amount of material, A is the amount at time t, and k is the decay constant. k is related to the half-life (t 1/2) by the following: (ln 2)/k=t. To get this equation, set A=1/2 of A(0) in the first equation, remove the A(0), take the natural log of both sides, and you're done.
There are lots of fun projects associated with a Geiger counter. Cosmic radiation is one (remind me to graph my week-long Ryerson graph). Live web server graph of current levels is another.
I have another graph of a more recent trip to LAX here.
41 comments:
I should note that on the airplane graph it looks like the radiation levels lower as you approach LA. If the plane were at the same altitude, the difference you are seeing is due to a change in geomagnetic latitude between Chicago and LA. The closer to the equator you are the fewer cosmic rays you encounter. And, the rays you do encounter are higher energy.
It even looks like a continuous slope downward. I wonder if we could best-fit that part of the flight, and graph latitude as a function of time.
Chicago is 52 degrees geomagnetic latitude; LA is 40. This is according to http://www.sec.noaa.gov/Aurora/
I should note I now think the uR/hr count on the airplane graph are too high--I think the AW-srad software thought it had a less sensitive detector installed.
Geiger counters measure ionisation. The basic idea is that most radiation (and some other things) will ionise the air and so you can detect that ionisation.
So, yes, your geiger counter will measure alpha particles (Helium nuclei), beta particles (high speed electrons and positrons) and Gamma radiation (photons).
You probably will find your half life is connected with the entire decay chain if it's fast enough, as the secondary decays are occurring while you are still watching. If you have a large half life somewhere in the chain then you will find that the obeserved half life is probably only the sum of the half lives above that one in the chain.
For example, the Uranium-238 -> Lead-206 chain has fast and slow decays.
U-238 -> Th-234 + Alpha (4,500,000,000 years)
Th-234 -> Pa-234 + Beta (24.5 days)
Pa-234 -> U-234 + Beta (1.14 minutes)
U-234 -> Th-230 + Alpha (233,000 years)
Th-230 -> Ra-226 + Alpha (83,000 years)
Ra-226 -> Rn-222 + Alpha (1,590 years)
Rn-222 -> Po-218 + Alpha (3.825 days)
Po-218 -> Pb-214 + Alpha (3.05 minutes)
Pb-214 -> Bi-214 + Beta (26.8 minutes)
Bi-214 -> Po-214 + Beta (19.7 minutes)
Po-214 -> Pb-210 + Alpha (0.00015 seconds)
Pb-210 -> Bi-210 + Beta (22 years)
Bi-210 -> Po-210 + Beta (5 days)
Po-210 -> Pb-206 + Alpha (140 days)
So if you're watching that decay for a few minutes (or hours) you are most likely to see the Po-218 -> Bi-210 section and that will go past in around 50 minutes. you might also see the Th-234 -> U-234 section.
Sources : Absolute Astronomy Geiger Counter information
Atral.com U-238 Decay chain
Web Elements Periodic table
I hope that this helps.
Z.
While a Geiger counter *could* measure betas (electrons) and alphas (helium nuclei), most won't. Geiger counters will detect ionizations that occur from gammas (high energy photons) or neutrons interacting with the material inside the chamber. The shielding on most counters prevents betas and alphas from interacting. They sometimes have windows (with protective coverings normally closed) to allow betas (primarily) to interact.
So, for your radion decay you most likely detected the gammas given off as part of the decay. Alphas are so weakly penetrating that a simple sheet of paper will stop almost all alphas.
While on the airplane, the earth's magnetic field is altered and atmosphere above the plane is diminished, allowing more cosmic radiation (gammas and neutrons) to make it to the counter, resulting in higher reads. This cosmic radiation is significant enough that most nuclear plant workers receive less radiation during normal operations than do aircraft personnel (and yet only the nuclear plant operators are considered "radiation workers")
This particular detector has a big mica window that will detect alphas and betas. You can see this when I measured a thorium-rich camera lens.
http://dwarmstr.blogspot.com/2005/03/pentax-takumar-lens.html
Er, that is http://dwarmstr.blogspot.com/2005/03/pentax-takumar-lens.html
Just a mention, but they use depleted uranium as the ballast in most commercial airliners now, and I think that's the deal, here. Do a google on "depleted uranium commercial airliners" and see for yourself. That was an issue with the twin towers collapse (breathing) and other 9/11 events that are not being publically discussed.
The radiation levels increase and decrease in direct relation to the airplane's altitude, so depleted uranium counterweights are out as the source.
Craig DeForest here -- just flitted over from BoingBoing.
The radon activity curve you saw from the filter will gradually converge to a single rate-limiting half-life: the half-life of the slowest decaying thing in the decay chain. Upstream nuclei with shorter half-lives will gradually decay away, and downstream nuclei will have their abundances set by the production rate from the slow step. Think of one of those cascade fountains where water gets poured from reservoir to reservoir: the reservoir with the smallest drain hole sets the rate for the whole chain under it.
If you're really using a Geiger counter then the software can make at best a rough guess at the amount of human dose. Geiger counters use a critical cascade to completely ionize the whole sensor tube when (ideally) even one gas molecule gets ionized. (free electrons gain enough energy to ionize other gas molecules, before they have traveled their mean free distance through the tube). Proportional counters use a lower applied voltage, so that the tube doesn't get fully ionized. The total charge pulse coming out of the tube is then proportional to the amount of energy deposited by each incoming particle of radiation. Proportional counters give you a better sense of the radiation dose you're getting, because they can account for the total amount of ionization from each incoming particle (which GM tubes can't).
If you want to get more sophisticated than that, you have to start thinking about radiation "quality factor", a multiplicative fudge factor on the total amount of ionization. QF estimates the biological effectiveness of each type of ionization -- gamma rays near 1MeV have a QF of 1, because they are the standard for biological dose, but alpha particles have a higher QF, because their ionization effects tend to be more concentrated in a small region -- so, on average, more of your cells will die with a certain amount of ionization from alpha particles than with the same amount of ionization from gamma rays. Then there are penetration effects -- alpha rays are much more effective, but external alpha rays (say from uranium ore) are usually completely stopped by your epidermis -- which is dead anyway.
So, er, if you have a GM tube that's merely counting pulses, the microrads/hour figures produced by the SW are just estimates; and if you want to figure human dose (microREM/hour, generally close to the microRAD/hour figure produced by your counter) it's even more of a guesstimate.
Very cool experiment, by the way. I had no idea anyone was making USB radiation counters -- gotta go get me one... :-)
I love the idea that you would buy a Geiger counter for no other reason then it is fun to learn about. My friends think I'm nuts because last year I bought myself an Oscilloscope and Microscope just to play with them.
Why would an airliner need balast? Trim tabs trim the aircraft up very nicely in flight. Carying that extra weight would simply result in increased fuel costs. With airlines pinching pennies anywhere they can, what would the benefit be?
So, what does this say about film (unexposed and exposed) and what it receives in flight vs what it gets in the carry-on x-ray machines in security?
A trim tab can cause drag and cost fuel as well. Might be worse than a little balast at certain speeds or alititudes.
Froskur - Make as much fun as you like, but the fact is that commercial airplanes use or have used DU as ballast. It's not ballast as what you would use for a seagoing vessel, but rather an opposing weight on a specific collection of parts to achieve a weight balance or imbalance.
Additionally, had you just performed a simple Google search as the original DU poster suggested, you could have found this out for yourself.
Check out Not-so-Depleted Uranium (about half way down the page under the 'Not-so-Depleted Uranium' heading).
I'll make even more fun of the DU guy.
I've fired DU rounds from an M1A1 tank, taken an "extreme" Nuclear, Biological, and Chemical (NBC) course, AND right before I left the Army they started giving ALL soldiers who may have a higher chance of coming into contact with DU special classes on DU fragments, inhalation of such fragments and whatnot. The SAME question was raised in a lot of that training about DU in airplanes.
The response?
"Not in civlian aircraft. Too risky." Of course, the Spectre carries a 105 main gun which is the same German-built gun in the older M1-series tanks AND is capable of firing DU rounds, but that would be expensive overkill I would think.
Now, if what you are saying is TRUE (I doubt it, no matter WHAT you "Google-find" on the Internet) then there would be radiation hazard teams at EVERY plane crash. Hmmm...I think the press would have picked up on that one since most of them are educated and would know a Geiger Counter when they saw it or asked "Hey, what is that?"
Hi all,
Not to put too fine a point on it, but the whole DU-counterweight-in-passenger-aircraft issue is completely overblown. Stop to think for one second - a 500 degree fire in the aircraft is going to be a much more immediate hazard to life than any amount of depleted uranium that might be emitted as a result.
As a certified industrial hygienist, I examine risks like this every day - "Where is the potential exposure?" is a question I ask frequently. Here, unless a catastrophic failure occurs, not even one photon of radiation from any DU is going to reach you, the passenger. Compare that with the cosmic radiation Dean observed with his toy G-M probe.
As for the DU ammunition stories that drift about the internet, I have no illusions that our government or military leaders have any hesitation to use weapons that present serious hazards to those who use them and those on whom they are used, But I haven't seen any evidence that even thousands of antitank rounds expended in Gulf War I can be tied to the horrifying natal deformities seen on some of the websites devoted to exposing this "crime". Even if we left hundreds of thousands of tons of DU in the deserts of Iraq, how are people going to be exposed to it? Where is the exposure? Are they burning it for heat and inhaling the fumes? In all likelihood, those deformities are caused by the starvation of the Iraqis facilitated by the US government.
They Don't Ballast Airplanes.
Ballast is used on ships, not planes. If they need to change the balance on an airliner they can shift gas between tanks. They also have little trim tabs on the wings that they can adjust to change the orientation of the plane in flight.
Airplanes cost more to fly as they get heavier. Manufacturers go to elaborate lengths to make planes as light as possible without making them weak. *Everything* that goes into a plane is scrutinized to see if it can be made lighter. Commercial airlines don't carry so much as one extra meal tray when they take off.
If for some bizarre reason they needed to make part of a plane heavier they would just use safe cheap lead weights, not expensive and hazardous uranium byproducts.
The exposure to the DU in Iraq comes from
the vaporization of it when the shells hit
tank armor. Very fine particles result
which settle out because of their density.
The dust gets stirred up in dust storms,
when kids play in tanks disabled by DU
rounds, and when traffic stirs up such
dust. The dust gets breathed, on people's
hands and on their foods.
A German doctor did a good study of this,
including taking radiation readings in Iraq.
Ed T says " Froskur - Make as much fun as you like, but the fact is that commercial airplanes use or have used DU as ballast.
and offers this link
"Check out Not-so-Depleted Uranium (about half way down the page under the 'Not-so-Depleted Uranium' heading)."
I did check this out, and the site refers to a plane crash in Holland where, apparently, signs of DU were found at the crash site.
It goes on to quote members of the American society for Metals( website here:http://www.asm-intl.org/)
without providing a link for the quote.
"The journal of the American Society for Metals explains that DU counterweights "are used in the aerodynamic controls of planes, rockets and helicopters to maintain the aircraft's center of gravity... in many civil and military aircraft."
Now, I don;t have access to the journal, but a simple search of the ASM site reveals no matches for 'depleted uranium"
It's hard to believe that they wouldn't have ever mentioned it at all if it was in their journal.
I smell urban legend.
Look, Occam's Razor says "planes do not need ballast."
even if we stipulate they do, I concur with the poster who suggest that far safer, nearly as dense metals would be used.
-Adam
I worked for the Boeing Co. from 1964 to 1966. One of my systems on the 727 was the Stall Warning system. As one of its inputs it used a Giannini made angle of attack indicator which had a small amount of DU as a counter-weight to balance the vane which was outside in the slip-stream. There were two of these sensors on each 727. The total mass was no more than a couple of ounces of Uranium. These vanes were located close to the cockpit.
Well, I've never taken a survey instrument on a commercial flight, but those numbers from the graph seem low to me. I'm in charge of the dosimetry where I work, and I've had guys accidentally take their dosimeters on flights. Those dosimeters have all turned up exposures in the 100-200 mr range for a cross-ocean flight. That's about a year's worth of exposure around here.
I suppose they could all have put their dosimetry through the x-ray bag scanner as well. Maybe you could try slipping your detector through the bag screener and recording that to see what happens.
I am an aerospace engineer.. Most of the previous comments about ballast on airplanes are very misguided if not completely wrong. Here's the REAL reason ballast is used on airplanes.
Yes, weight DOES matter. However, what is just as important is drag (the resistance to moving thru the air). An airplane MUST have its center of gravity closely maintained so that it will be stable in flight - so it will not tend to diverge from controlled flight. Ballast is absolutely required for this. Additionally, while an airplane can be safely flown within a range of center of gravity locations, it will require some aerodynamic "trim" to keep it moving straight. This is usually done by adjusting the control surfaces. However, using controls to adjust trim adds drag - which means more fuel is needed for the engines to overcome the drag.
Many modern jetliners use fuel shifting to accomplish this center of gravity trim. So ballast is not needed dynamically - only when it is built. Bear in mind that most of the time, the passengers and baggage cause huge changes in balance, that must be countered.
Now, this is almost all beside the point anyway. The ballast being discussed here is really not related to balancing the center of gravity - it's related to balancing individual control surfaces on the plane. To prevent a control surface from entering a "flutter" state - where it violently oscillates back and forth, and potentially self-destructs - the control surface must have its OWN center of gravity balanced. Obviously a control surface is not a huge place to mount weights wherever you wish - so the denser the ballast material, the better. So this is the place in the airplane where DU is frequently used. It's not that large a quantity, and it's not in a location that is likely to be released, other than a crash of course.
A summary of exposure rates is available at Radiation Exposure During Commercial Airline Flights. In short, they found rates of 200-300microREM/hr for flights with similar geomagnetic latitude.
i was always led to believe that the danger from DU (please someone not the "depleted" in the title) comes from the toxicity of uranium. Its a far more toxic metal than either cadium or lead especially as a pentoxide i believe. i think it does have teratogenic effects but only if the MOTHER (not the father) is exposed in the first couple of weeks of pregnancy (when she doesn't realise she's pregnant) and as far as i know there's no bioconcentration of uranium in the human body, much like mercuric chloride that passes straight through biological membranes without resistence so can't be sequested.
DU-guy...
Boeing and McDonnell-Douglas discontinued using DU as ballst in the 80s - and guess the manufactures of the "WTC-planes"..?
I agree with john roper. I think it it totally cool that Dean is "learning by playing." I collect all kinds of unusual high-end technical equipment including Geiger counters, microscopes, oscilloscopes, radio gear, gas lasers, etc. I have learned from everything I've aquired. I like being the "only kid on my block" who has whatever oddity I have come across. I have also learned a lot from this thread. Dean, you've got the spark! Kudos!
As for the UD in airliners... If it is used, I doubt that there is any real risk--the whole thing seems over-blown. All you have to say is "radiation" and the hackles flair up on every conspiracy theorist. Don't believe everything you read, even if there is a slash of truth to it. Think critically!!!!
Sorry... That was "splash of truth" not "slash."
very interesting blog dean! I'm off to Pripyat as soon as the snow melts then on to reactor no 4 to make a short film .walking around chernobyl without a gieger counter is like walking in a mine field with snowshoes on! It did cross my mind about checking doses on the plane but I thought that was silly. I might try it on the way home now I know i'm not the only one! Don't think i'll try it on the way there as I don't want it confiscated on the way as I think I may be needing it!
Great information. You have a nice way of writing so that everyone can understand what you are saying.
Is this for earthquake. Do you have any news about Hitti?
Geiger counters only measure ionizing radiation. For earthquakes you need a seismometer or seismograph.
I used to have to hump lead ballast every time we installed or removed our equipment in a high altitude research aircraft. This was only necessary so that acceptable trim could be maintained without manual outside adjustments to control surfaces. Cargo aircraft like the C130 have internally adjustable trim wheels. In one aircraft the nose bay ballast was normally lead but could be substituted with a cooler containing seafood from Alaska.
Know your facts!
http://iopscience.iop.org/0952-4746/22/1/602
U-238 low fissible counter weight trim tabs
ok lets see my Gieger Counters https://www.cdhpl.com/best-geiger-counter/
HealthFusion Login - Are you getting trouble while log into your account, No issue, Here is the complete step by step technique. Healthfusion Login
TuTuApp is just an amazing app store from where you can download everything for free. TuTuApp is available for Android and iOS devices. TuTuApp
If you are looking for the complete procedure of MyAARPMedicare Login guide and rewards, forgot the password, contact details, Plans, phone numbers and other things. www myaarpmedicare com
Amazing Post, Thank you for sharing this post really this is awesome and very useful.
1337xunblock
Post a Comment